

 |
 |
Comp Chem Research Developments | |
| Archive of Comp Chem Research News | |
|
Dec 12, 2002 |
|
|
|
Although most chemical reactions can be understood in terms of thermally activated molecules crossing barriers on the Born-Oppenheimer potential energy surface, it is well known that quantum mechanical tunneling "through" barriers is sometimes more important than passage "over" barriers. However, tunneling is also thermally activated under most conditions. It is well known that quantum mechanical tunneling can play an important role in reactions in which a hydrogen atom, proton, or hydride ion is transferred. Such tunneling contributions to rates have recently been implicated in reactions as diverse as those in interstellar space, enzyme catalysis, and organic chemistry. As the temperature is lowered, the reactant population becomes concentrated in the lowest-energy quantum state; and one should, in principle, be able to observe tunneling out of this single vibrational state. Peter S. Zuev and Robert S. Sheridan of the University of Nevada, David A. Hrovat and Weston Thatcher Borden University of Washington, and postdoctoral associate Titus V. Albu and Professor Donald G. Truhlar of our department have reported an observation of this single quantum-state limit for a reaction involving carbon tunneling. They observed ring expansion of 1-methylcyclobutylfluorocarbene at 8 K, a reaction that involves carbon tunneling. At this temperature the limiting reaction rate is 4.0 x 10-6 s-1; and the tunneling contribution to the rate is calculated to be 152 orders of magnitude greater than the contribution from passing over the barrier. The reaction proceeds from a single quantum state of the reactant, and they also reported environmental effects of the solid-state, inert-gas matrix on the reaction rate. The theoretical rate calculations were based on methods developed at the University of Minnesota, in particular 1-parameter hybrid density functional theory for kinetics and variational transition state theory with multidimensional tunneling contributions. |
|
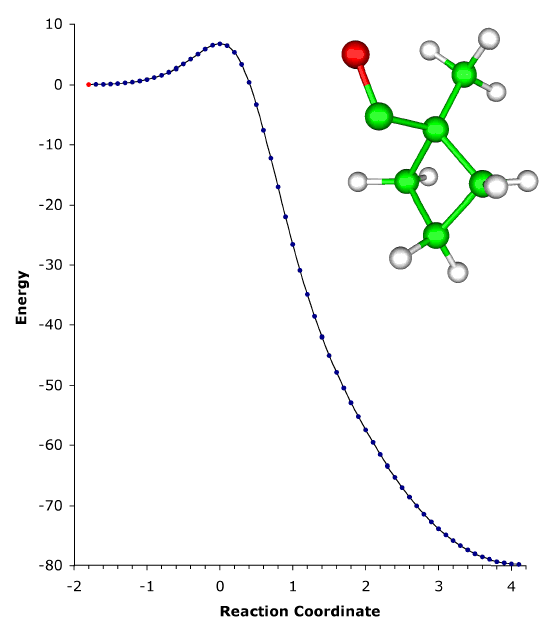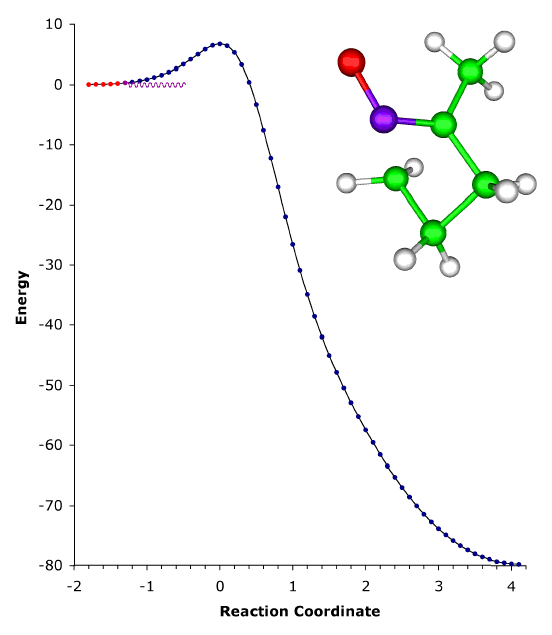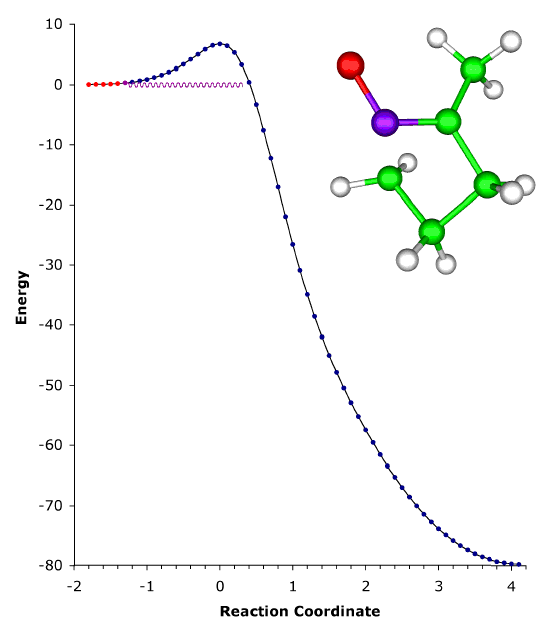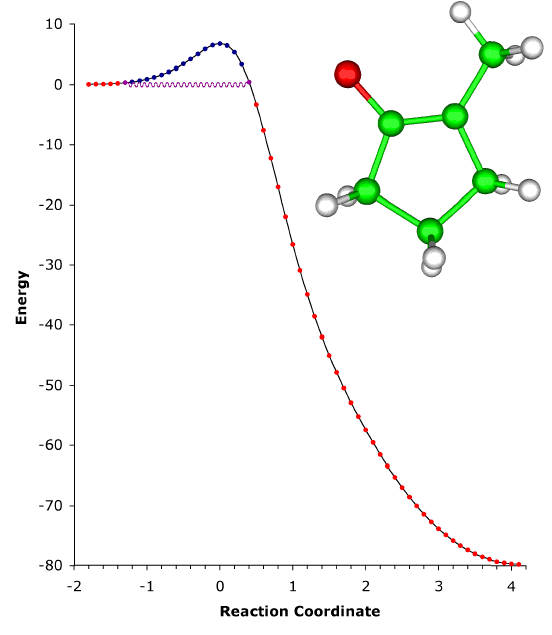
ABOVE: The ring expansion of 1-methylcyclobutylfluorocarbene in a solid-state matrix at 8 K has been observed and interpreted, and the rate constant has been measured and calculated [P. S. Zuev, R. S. Sheridan, T. V. Albu, D. G. Truhlar, D. A. Hrovat, and W. T. Borden, Science 299, 867-870 (2003)]. The reaction is remarkable in two respects: (1) It occurs almost entirely (actually, 9.99...5%, where ... denotes 148 more 9s) by tunneling, even though the reaction coordinate is dominated by movement of carbon. (Usually tunneling is observed only for light atoms like hydrogen and deuterium.) (2) The reaction rate has reached its low-temperature limit, corresponding to reaction from a single quantum state; this limit plays a key role in determining the shape of Arrhenius plots at low temperature, but it has rarely been observed. The movie shows the geometry changes that occur along the reaction pathway from 1-methylcyclobutylfluorocarbene to 1-fluoro-2-methylcyclopentene. For the portion of the reaction pathway in which the reactant is tunneling through the barrier that separates it from the product, the carbenic carbon turns purple. The figure to the left of the molecule shows the interpretation of the process in terms of movement governed by an effective potential energy curve. |
|
|
BELOW: Four snapshots along the reaction path. The snapshots correspond to geometries along the vibrationally adiabatic ground-state potential curve. Two key bond lengths are indicated in angstroms. 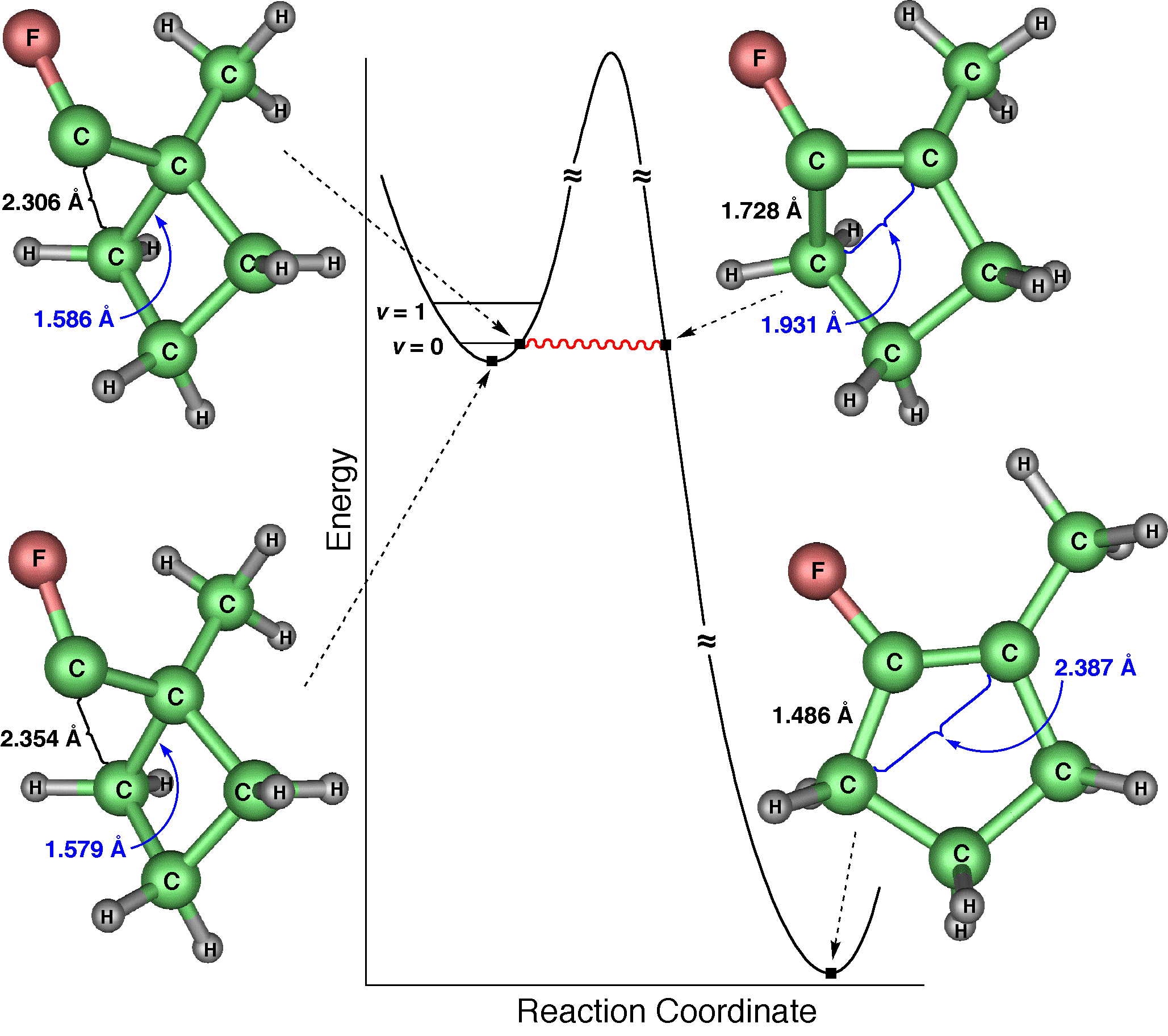
|
|
|
|