

 |
 |
Comp Chem Research Developments | |
| Archive of Comp Chem Research News | |
August 8, 2001
|
|
|
|
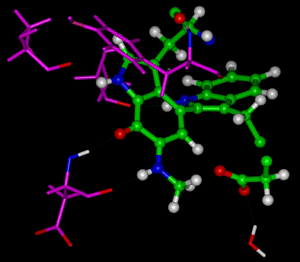
The catalytic power of enzymes is remarkable. For example, the enzyme orotidine 5’-monophosphate decarboxylase enhances the rate of decarboxylation of orotic acid by an astonishing 1017 fold over that in aqueous solution. The fundamental question is what are the major factors that contribute to enzyme’s catalytic efficiency. The classic picture of transition state stabilization, in which enzyme binds preferentially to the species near the top of the activation barrier and consequently lowers the free energy of activation, is widely invoked. Alternatively, the binding energy of the substrate can be utilized by the enzyme to destabilize the ground state, leading to a reduction of the barrier to reaction. The specific means by which the activation barrier is reduced remains vigorously debated. Another way to effectively lower the effective threshold energy for reaction is quantum mechanical tunneling. In the past decade, strong experimental evidence for quantum mechanical tunneling in enzymatic reactions has been accumulated for a number of enzymes. The experimental evidence was primarily derived from kinetic isotope effects (KIEs). Although interpretation of the experimental data is often difficult due to the need for making mutations to unmask the rate-limiting step, recent theoretical calculations in the research groups of Professors Jiali Gao and Donald Truhlar employing multidimensional tunneling approximations have fully confirmed the importance of quantum mechanical tunneling effects in enzyme catalysis ["Quantum Dynamics of Hydride Transfer in Enzyme Catalysis," C. Alhambra, J. C. Corchado, M. L. Sanchez, J. Gao, and D. G. Truhlar, Journal of the American Chemical Society 122, 8197-8203 (2000)]. These researchers have developed a theoretical procedure for including quantum mechanical effects in enzyme kinetics that features a combination of molecular dynamics simulations and variational transition state theory. The method was applied to the proton transfer reaction catalyzed by methylamine dehydrogenase (MADH), which converts primary amines into aldehydes. The computations reveal that the effect on the free energy of activation by quantizing bound vibrations lowers the barrier by 3.2 kcal/mol. Quantum mechanical tunneling effects further lowers the free energy of activation by 2.5 kcal/mol, giving rise to a total lowering of DGact due to quantum effects that equals 5.7 kcal/mol. From the computed proton and deuteron transfer rate constants, which include multidimensional quantum tunneling contributions, these researchers["Quantum Mechanical Tunneling in Methylamine Dehydrogenase," C. Alhambra, M. L. Sánchez, J. C. Corchado, J. Gao, and D. G. Truhlar, Chemical Physics Letters, to be published] obtained a computed KIE of 18.3 for the proton abstraction reaction in MADH, which may be compared with the experimental value of 17.2 ± 0.6. Note that conventional transition state theory without tunneling yields a considerably smaller KIE, in particular 5.9. The calculations indicate that about 1% of the reaction occurs by overbarrier processes, with the rest due to tunneling. This provides the most striking evidence yet for the contribution of tunneling processes in enzymatic reactions at physiological temperatures. |
|
|
|
|
|